FDA Breakthrough Device for Early and Non-Invasive Urothelial Cancer Recurrence Monitoring
Overview
TAGMe™ (Tumor Aligned General Methylated Epiprobe) is a proprietary DNA methylation–based assay designed for non-invasive detection and longitudinal monitoring of urothelial carcinoma (UC) using urine samples.
Built on advanced epigenetic marker discovery and high-throughput methylation quantification, TAGMe enables earlier detection, improved sensitivity, and dynamic monitoring of recurrence, addressing the critical unmet need in post-treatment surveillance.
TAGMe DNA Methylation Detection Kits (qPCR) for Urothelial Cancer are CE-marked products for In Vitro Diagnostic Use. The TAGMe DNA Methylation Detection Kits (qPCR) for Urothelial Cancer kit is not FDA-cleared.
Clinical Need
Urothelial carcinoma is characterized by:
High recurrence rates (up to 50–70%)
Requirement for frequent lifelong surveillance
Heavy reliance on cystoscopy, which is:
Invasive and uncomfortable
Operator-dependent
Cost-intensive with repeated procedures
Urine cytology, while non-invasive, suffers from low sensitivity, especially for early-stage disease
There is a clear clinical demand for a non-invasive, sensitive, and scalable monitoring solution.
TAGMe has been granted U.S. FDA Breakthrough Device Designation for the detection and monitoring of urothelial carcinoma.
This designation is awarded to technologies that:
Provide more effective diagnosis or monitoring of life-threatening diseases
Address unmet clinical needs
Demonstrate potential for significant clinical advantage
Implications:
Priority interaction and guidance from FDA
Accelerated development and review pathway
Strong validation of clinical and technological innovation
This positions TAGMe as a next-generation standard candidate in non-invasive urothelial cancer surveillance
(Breakthrough Device Designation does not mean FDA clearance or approval)
Technology Platform
TAGMe™ Epigenetic Marker System
Derived from genome-wide methylation discovery (GPS technology)
Represents a pan-cancer epigenetic marker database
Enables:
Tumor-specific signal detection
High signal-to-noise discrimination
Early-stage tumor identification
Bisulfite-Free Me-qPCR Platform
Unlike conventional methylation assays, TAGMe utilizes a bisulfite-free methylation detection approach, enabling:
Faster workflow (≈ 4.5 hours total)
Higher throughput (up to 280+ samples/day/operator)
Reduced DNA degradation
Simplified laboratory workflow
Clinical Performance
Overall Performance
Sensitivity: ~85%
Specificity: ~94%
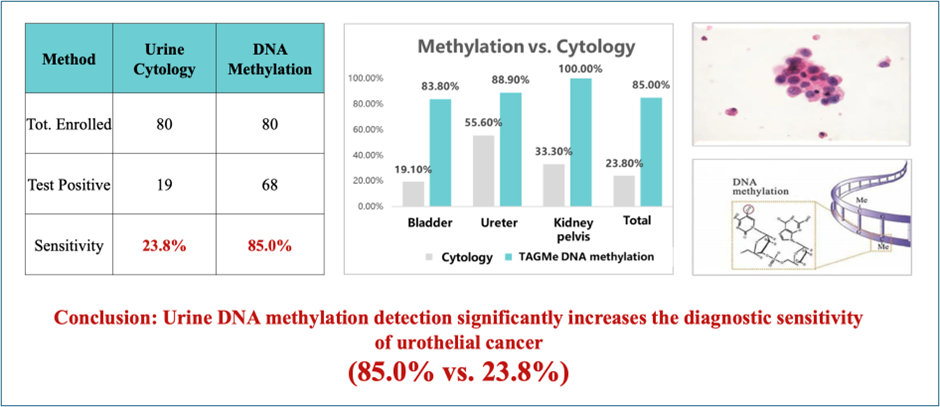
Compared to Urine Cytology
TAGMe: 85.0% sensitivity
Cytology: 23.8% sensitivity
Early-Stage Detection
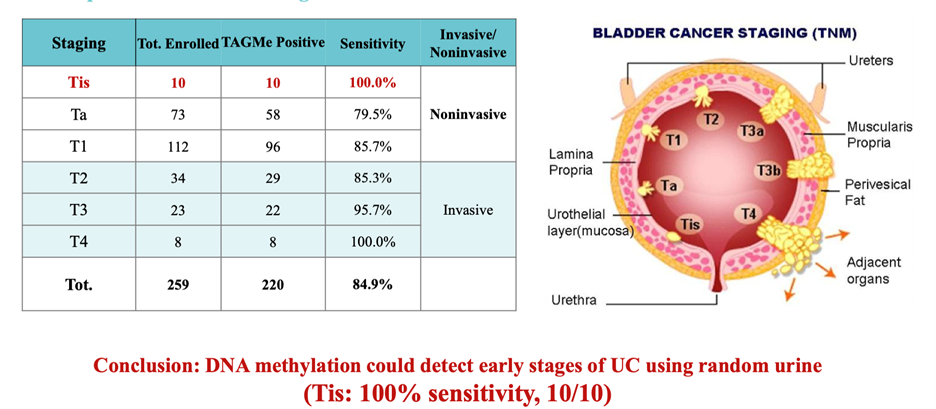
Carcinoma in situ (Tis): 100% sensitivity (10/10)
Strong performance in non-muscle-invasive bladder cancer (NMIBC; pTis, pTa, and pT1), the key population for recurrence monitoring.
Recurrence Monitoring Capability
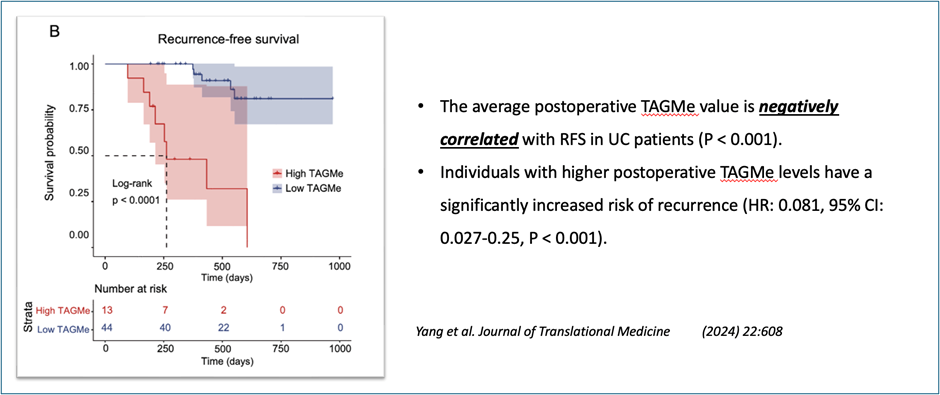
TAGMe is a longitudinal monitoring system:
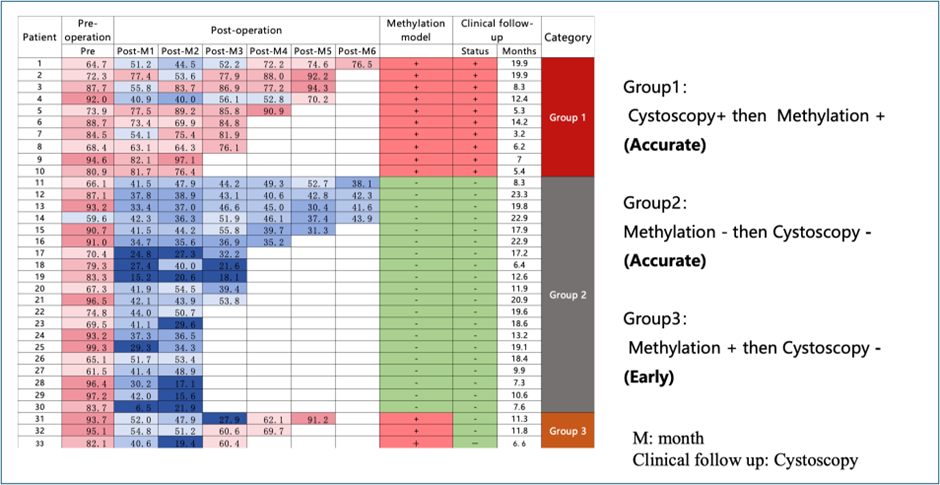
Recrudesce monitoring model—Early prediction of cancer recurrence
Dynamic Quantitative Monitoring
Provides continuous methylation score tracking over time
Enables:
Early recurrence detection
Risk stratification (negative → weak → high positive)
Treatment decision support
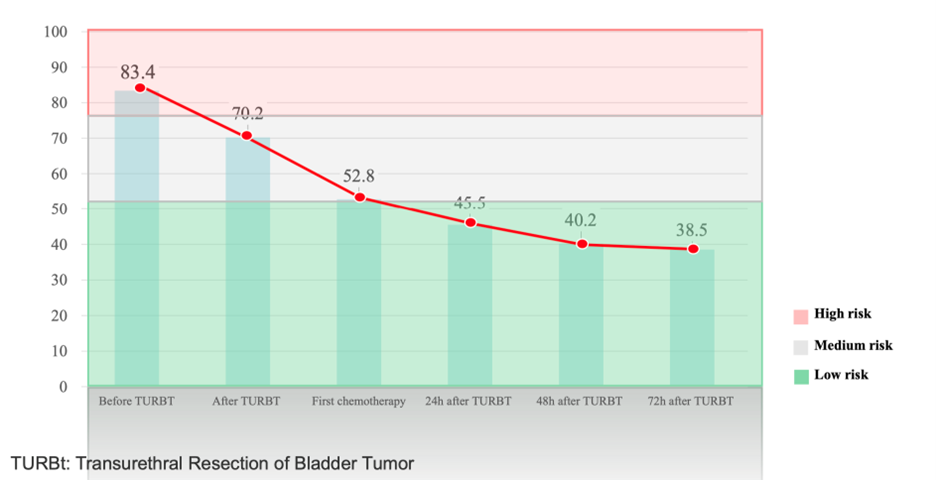
Dynamic monitoring——Solution for full process of UC
Early Detection vs. Cystoscopy
Detects recurrence 3–15 months earlier than cystoscopy
Clinical Utility
Identify recurrence earlier
Reduce unnecessary invasive procedures
Optimize follow-up intervals
Improve patient compliance
Workflow & Sample Handling
Sample type: Urine (30 mL, random or morning sample)
Storage: Room temperature up to 7 days
Total turnaround time: ~3 hours (PCR-based workflow)
Simple Workflow
Urine collection
DNA extraction
Me-qPCR detection
Automated analysis & report
Clinical Applications
TAGMe supports a full continuum of UC management:
1. Recurrence Monitoring (Primary Use Case)
Post-TURBT surveillance
High-frequency longitudinal testing
Early recurrence detection
2. Adjunct to Cystoscopy
Reduce unnecessary procedures
Improve diagnostic confidence
3. Treatment Response Monitoring
Evaluate surgery or chemotherapy efficacy
4. Risk Stratification
Identify high-risk patients requiring closer follow-up
Patient-Centered Advantages
Non-invasive (urine-based)
High compliance for repeated testing
At-home or clinic collection
Reduced procedural burden
Laboratory & Commercial Advantages
High-throughput compatible with CLIA labs / LDT model
Short turnaround time → supports rapid clinical decision-making
Lower operational complexity vs. bisulfite sequencing
Scalable for central lab or distributed testing models
Ordering Info:
Cat. No. | Description | Size |
10048 | TAGMe DNA Methylation Detection Kits (qPCR) for Urothelial Cancer | 48-reactions |
TAGMe DNA Methylation Detection Kits (qPCR) for Urothelial Cancer kit is a CE marked product, and for In Vitro Diagnostic Use.
TAGMe DNA Methylation Detection Kits (qPCR) for Urothelial Cancer kit is not FDA-cleared.
Reference:
1. Yang Z, Chen Q, Dong S, Xu P, Zheng W, Mao Z, Qian C, Zheng X, Dai L, Wang C, Shi H, Li J, Yuan J, Yu W, Xu C. Hypermethylated TAGMe as a universal-cancer-only methylation marker and its application in diagnosis and recurrence monitoring of urothelial carcinoma. J Transl Med. 2024 Jul 2;22(1):608. doi: 10.1186/s12967-024-05420-3. PMID: 38956589; PMCID: PMC11218302.