One Sample. Earlier Answers. Broader Coverage.
The EpiOnco Gynecologic Cancer Test is a next-generation DNA methylation assay designed to advance women’s cancer detection beyond conventional one-disease-at-a-time screening. Built on a GPS marker-discovery engine and bisulfite-free Me-qPCR platform, it is designed to detect cervical cancer, cervical adenocarcinoma including gastric-type adenocarcinoma, and endometrial cancer from a single cervical sample, with the potential to identify disease risk as early as the precancerous stage.
A More Advanced Approach to Gynecologic Cancer Detection
Current screening pathways have improved outcomes, but important clinical gaps remain.
HPV testing is highly useful, yet HPV positivity is not equivalent to cancer, often creating excess follow-up, unnecessary anxiety, and referral burden. Cytology remains an important tool, but it is inherently dependent on specimen quality, interpretation, and access to experienced pathology resources. These limitations are especially relevant for glandular disease, including cervical adenocarcinoma, where missed detection remains a real concern. Endometrial cancer adds another challenge: there is still no widely adopted, accurate, minimally invasive screening solution for routine early assessment.
The EpiOnco Gynecologic Cancer Test was developed to address these unmet needs with a platform that is molecularly objective, operationally streamlined, and clinically actionable.
What This Test Covers
From one cervical collection, the assay is designed to support risk assessment for:
Cervical squamous cell carcinoma
Cervical adenocarcinoma
Gastric-type cervical adenocarcinoma
Endometrial cancer
High-grade precancerous lesions associated with these diseases
This broader clinical scope helps address a major weakness of conventional workflows: fragmented testing across multiple gynecologic cancers.
Why EpiOnce Stands Out
Multi-cancer insight from a single sample
Rather than screening one condition at a time, EpiOnce is positioned as a single-sample molecular solution for multiple clinically important gynecologic cancers. That means a more efficient workflow for laboratories and a more comprehensive answer for clinicians and patients.
Earlier signal warning
DNA methylation changes arise early in carcinogenesis and can remain informative across the disease continuum, from early risk detection to treatment monitoring and recurrence assessment.
Bisulfite-free methylation detection
The assay is built on Me-qPCR platform, a bisulfite-free methylation workflow designed to reduce complexity, preserve DNA integrity, and improve reproducibility compared with conventional bisulfite-based workflows.
Quantitative, not just qualitative
Our methylation platform incorporates a proprietary quantitative analysis framework and dynamic monitoring model, enabling a more nuanced interpretation of disease risk rather than a simple yes/no result.
Designed for real clinical bottlenecks
The test is particularly relevant where existing screening pathways struggle:
High-risk HPV-positive triage
ASC-US and LSIL triage
Suspected glandular lesions
Post-treatment surveillance
Clinical Evidence
Strong discrimination for clinically meaningful cervical disease
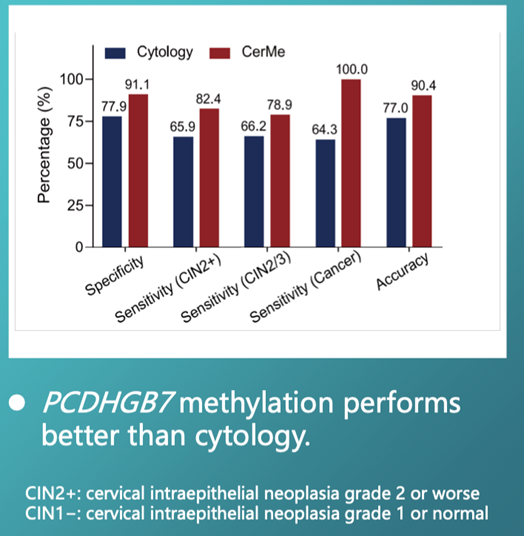
In published data, PCDHGB7 methylation showed clear separation between CIN2+ and CIN1− groups, with an ROC AUC of 0.91 (95% CI, 0.86–0.95), outperforming cytology in this setting.
Better triage performance in hrHPV-positive women
In a cohort of 3,251 high-risk HPV-positive women, methylation-based triage reduced both over-referral and missed diagnosis compared with cytology-based triage:
HPV + cytology triage: over-referral 20.58%, missed diagnosis 6.50%
HPV + methylation triage: over-referral 9.26%, missed diagnosis 3.24%
This is a meaningful advantage for healthcare systems seeking to improve colposcopy efficiency without sacrificing clinical sensitivity.
Improved support for adenocarcinoma detection
Among confirmed cervical cancer cases in hrHPV-positive women, methylation testing showed stronger sensitivity than cytology, particularly for adenocarcinoma, a subtype that remains more difficult to detect using conventional screening alone. We report missed detection rates of 0% vs. 21.9% for SCC and 0% vs. 40% for adenocarcinoma in the compared setting.
Potential value in gastric-type adenocarcinoma
This matters because gastric-type cervical adenocarcinoma is one of the most challenging cervical malignancies in routine practice. We report performance in this subgroup of 83.33% sensitivity, 86.96% specificity, 62.50% PPV, 95.24% NPV, and 86.21% accuracy.
Endometrial cancer detection from minimally invasive sampling
The platform also addresses a key unmet need in women’s health: earlier evaluation for endometrial cancer. In blinded studies, performance was reported as:
Pap brush samples: sensitivity 80.65%, specificity 82.81%
Tao brush samples: sensitivity 61.29%, specificity 95.31%
That creates an opportunity to expand beyond cervical-only workflows toward broader gynecologic cancer assessment.
Platform Strength Behind the Product
The product story is stronger because the platform story is stronger.
100+ collaborating hospitals
200,000+ double-blind validated clinical samples
18 PCT applications
17 invention patents
11 international authorizations
42 accepted patent filings
Its discovery engine is based on Guide Positioning Sequencing (GPS), which is a high-coverage methylome platform with 96% coverage, supporting broad marker discovery and full-lifecycle cancer monitoring.
This is not a one-off assay. It is a scalable epigenomics platform with translational depth.
Operational Advantages for Labs and Health Systems
The Me-qPCR workflow is positioned for real-world implementation:
Bisulfite-free
High reproducibility
Lower workflow burden
Faster turnaround
Quantitative readout for follow-up
The platform processed up to 282 samples per day on a single qPCR instrument with a single operator, compared with materially lower throughput for conventional bisulfite-based workflows.
That makes the test particularly attractive for:
reference laboratories
hospital molecular labs
women’s health screening programs
decentralized clinical settings seeking advanced oncology diagnostics without NGS-style workflow burden
Intended Use Scenarios
The EpiOnco Gynecologic Cancer Test is designed to support several clinically relevant scenarios:
1. Screening
For women seeking a more advanced molecular screening option, especially when broader gynecologic cancer coverage is desired.
2. Triage in hrHPV-positive patients
To improve referral efficiency and reduce unnecessary colposcopy burden.
3. Adjunctive evaluation in equivocal or high-risk cases
Including:
• ASC-US
• LSIL
• persistent symptoms despite negative routine testing
• suspicion of gastric-type cervical adenocarcinoma4. Post-treatment monitoring
The quantitative methylation framework supports longitudinal assessment after treatment and during surveillance.
A Stronger Vision for Women’s Cancer Detection
The future of gynecologic oncology diagnostics will not be built on fragmented, single-endpoint tests alone. It will be built on platforms that are:
earlier detection
clinically broader
operationally scalable
quantitative over time
fit for real-world implementation
The EpiOnco Gynecologic Cancer Test is positioned at that intersection—bringing together epigenomic discovery, multi-cancer insight, and clinical practicality in a single solution.
Bring a more advanced molecular strategy to gynecologic cancer detection. Contact us to discuss clinical adoption, laboratory partnerships, licensing, or regional commercialization opportunities.
Ordering Info:
Cat. No. | Description | Size |
20048 | DNA Methylation Detection Kits (qPCR) for PCDHGB7 | 48-reactions |
DNA Methylation Detection Kits (qPCR) for PCDHGB7 are CE-marked products for In Vitro Diagnostic Use.
DNA Methylation Detection Kits (qPCR) for PCDHGB7 are not FDA-cleared.
Reference:
1. Cao D, Yang Z, Dong S, Li Y, Mao Z, Lu Q, Xu P, Shao M, Pan L, Han X, Yuan J, Fan Q, Chen L, Wang Y, Zhu W, Yu W, Wang Y. PCDHGB7 hypermethylation-based Cervical cancer Methylation (CerMe) detection for the triage of high-risk human papillomavirus-positive women: a prospective cohort study. BMC Med. 2024 Feb 5;22(1):55. doi: 10.1186/s12916-024-03267-5. PMID: 38317152; PMCID: PMC10845746.
2. Yuan J, Mao Z, Lu Q, Xu P, Wang C, Xu X, Zhou Z, Zhang T, Yu W, Dong S, Wang Y, Cheng W. Hypermethylated PCDHGB7 as a Biomarker for Early Detection of Endometrial Cancer in Endometrial Brush Samples and Cervical Scrapings. Front Mol Biosci. 2022 Jan 4;8:774215. doi: 10.3389/fmolb.2021.774215. PMID: 35059435; PMCID: PMC8763697.